Scientists confirm: This is the most effective way to get your cat’s attention, according to new research
Elderly Couple Refuses Reserved Seats—Viral Train Standoff Sparks Fiery Debate on Courtesy
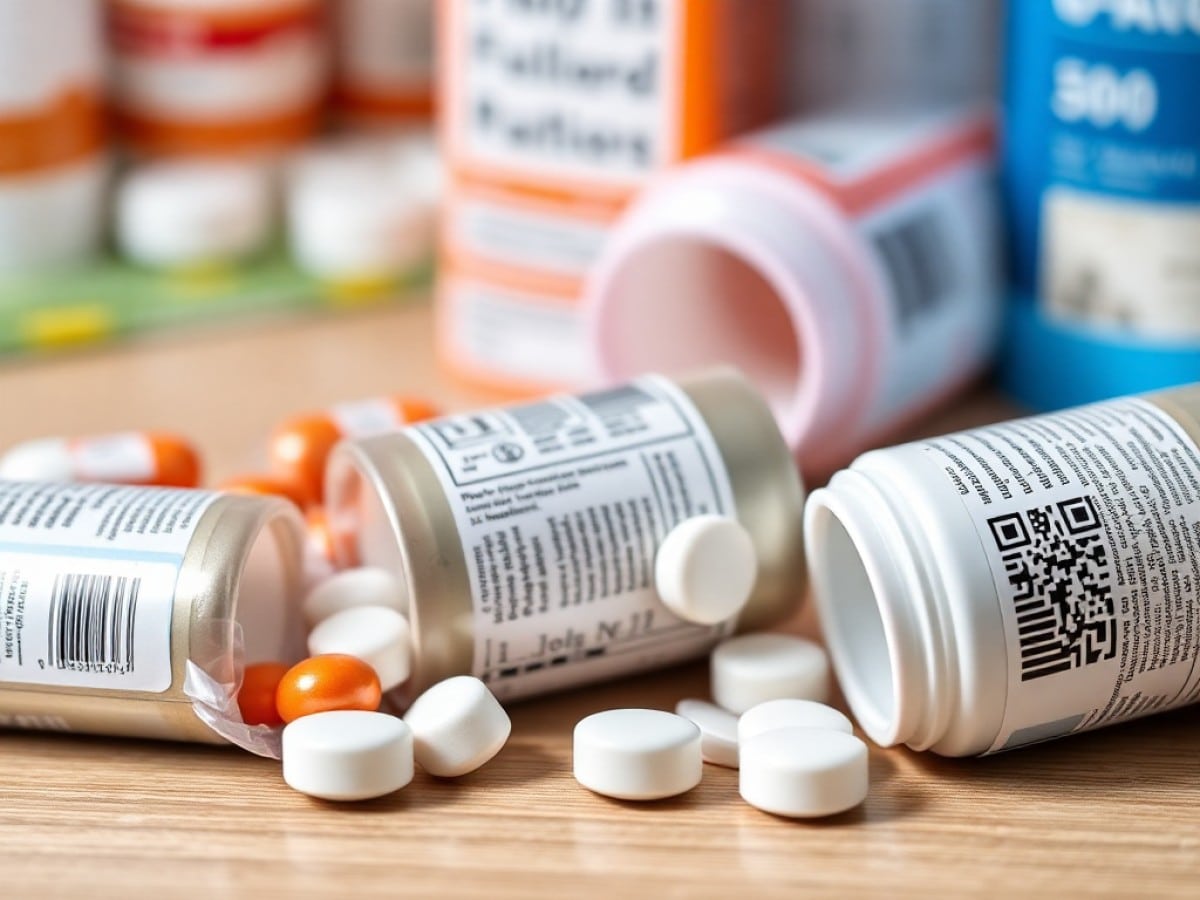
The digital transformation is now reaching into the realm of medication instructions. The National Agency for the Safety of Medication has initiated a large-scale trial wherein over 500 medication packages will include a QR Code. This code links to a digital version of the medication’s instructions. The trial has already been underway for several weeks and is scheduled to last two years. The aim is to assess how useful and accessible this digital approach is for both patients and healthcare professionals.
A QR code printed on the package provides easy access to an electronic notice (e-notice) hosted on the Public Database of Medications. The introduction of e-notices is intended to be gradual, with paper versions still included in medication boxes sold at pharmacies. In hospitals, however, some paper notices have been entirely replaced with digital versions as hospitalized patients rarely have access to paper notices.
While the shift to digital can pose challenges for certain groups, such as the elderly or those with disabilities, it also offers several benefits. Not only does it reduce the ecological footprint associated with producing medication, but digital notices can also be updated remotely to reflect the most current information. Additionally, they can be enhanced with videos or extra content such as safety information and thematic reports.
Of the 590 medications included in this testing phase, 420 are distributed in hospitals. These include cancer treatments, gene and cellular therapy medications, antibiotics, and antihypertensives. The remaining 170, available in pharmacies, include statins, vaccines, and various forms of paracetamol.
The objective of this trial is to gather data on how accessible and useful these e-notices are perceived to be. Two surveys will be conducted midway through the trial, and a satisfaction questionnaire will be implemented. Afterward, an evaluation will be conducted in collaboration with patient associations, healthcare professionals, and industrial partners. While France is not the only country exploring this transition, the European Commission has been advocating for a shift towards electronic notices, with countries like Belgium and Spain also running similar experiments.
iPhone and smartphone users have long had the ability to access digital medication instructions through apps that scan the datamatrix codes found on all medication boxes. Examples include the Medico app and the Medication Notices app, which relies on content provided by the National Agency for the Safety of Medication and Health Products.
Similar Posts
- Vital Card App Now Activates Without France Identité: Expanded to More Departments!
- Final Cut Pro & Logic Pro: Grab Your Last Chance for a Free 90-Day Trial!
- Vitale Card on iPhone: Now Available for All Users!
- GPT 4.1 Update: New Developer Model Now Integrated in ChatGPT!
- Claude Unveils Skills: Revolutionize Custom Instruction Manuals Creation!
Why You Should Never Reheat These Foods in the Microwave – The Hidden Dangers Experts Warn About
I tried the top 5 guard dogs—here’s what makes these breeds the ultimate protectors

Ava Blake is passionate about social media platforms and how they influence modern tech culture. She reviews apps, trends, and the evolving digital lifestyle for Touch Reviews readers.